Volume 6, Issue 2 (2025)
J Clinic Care Skill 2025, 6(2): 59-64 |
Back to browse issues page
Article Type:
Subject:
Ethics code: IR.YUMS.REC.1402.154
History
Received: 2025/03/20 | Accepted: 2025/05/8 | Published: 2025/05/12
Received: 2025/03/20 | Accepted: 2025/05/8 | Published: 2025/05/12
How to cite this article
Hashemimohammadabad Z, Hashemimohammadabad M, Yazdanpanah I, Razmjoue D, Karimi F, Hashemimohammadabad N. Effect of Melissa Officinalis on Depression in affected Individuals. J Clinic Care Skill 2025; 6 (2) :59-64
URL: http://jccs.yums.ac.ir/article-1-397-en.html
URL: http://jccs.yums.ac.ir/article-1-397-en.html
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



Rights and permissions
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Authors
Z. Hashemimohammadabad1 
 , M. Hashemimohammadabad2
, M. Hashemimohammadabad2 
 , I. Yazdanpanah1
, I. Yazdanpanah1 
 , D. Razmjoue3
, D. Razmjoue3 
 , F. Karimi4
, F. Karimi4 
 , N. Hashemimohammadabad *5
, N. Hashemimohammadabad *5 


 , M. Hashemimohammadabad2
, M. Hashemimohammadabad2 
 , I. Yazdanpanah1
, I. Yazdanpanah1 
 , D. Razmjoue3
, D. Razmjoue3 
 , F. Karimi4
, F. Karimi4 
 , N. Hashemimohammadabad *5
, N. Hashemimohammadabad *5 

1- College of Medicine, Zhejiang University of Medical Sciences, Zhejiang, China
2- College of Medicine, Zhengzhou University of Medical Sciences, Zhengzhou, China
3- Medicinal Plants Research Center, Yasuj University of Medical Sciences, Yasuj, Iran
4- College of Medicine, Yasuj University of Medical Sciences, Yasuj, Iran
5- Department of Psychiatry, College of Medicine, Yasuj University of Medical Sciences, Yasuj, Iran
2- College of Medicine, Zhengzhou University of Medical Sciences, Zhengzhou, China
3- Medicinal Plants Research Center, Yasuj University of Medical Sciences, Yasuj, Iran
4- College of Medicine, Yasuj University of Medical Sciences, Yasuj, Iran
5- Department of Psychiatry, College of Medicine, Yasuj University of Medical Sciences, Yasuj, Iran
Full-Text (HTML) (961 Views)
Introduction
Depression is a common psychiatric disorder and one of the most disabling mental health issues in various societies [1]. It is characterized by a mental state of low mood and reduced activity, affecting over 280 million people (about 3.5% of the world’s population). Classified as a mental and behavioral disorder, depression impacts the way people think, act, and feel. The main symptoms of depression may include a lack of interest and enjoyment in life (anhedonia), a mood disorder, or both [2]. Depression is a serious condition in today’s society, with one in 10 patients suffering from major depression [3].
The most commonly prescribed antidepressants are selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and tricyclic antidepressants (TCAs). SSRIs are among the newer treatments for major depression and are typically used as a first-line treatment. Escitalopram is one of these medications, prescribed in daily doses ranging from 10 to 60mg [4].
Antidepressants can take several weeks to show effects and may be accompanied by side effects. Some patients may struggle to adhere to medication due to these side effects or fear of using chemicals, which can lead to ineffective treatment [3].
Plants have been an important source of medicine for thousands of years. The World Health Organization estimates that nearly 80% of people still primarily rely on traditional remedies, such as herbs, for their medicines. Plants are also the source of many modern medicines. It is estimated that up to one quarter of prescribed drugs contain plant extracts or active ingredients obtained from plant substances [5].
Dracocephalum kotschyi Boiss possesses a number of medicinal properties, including antioxidant, antibacterial, anticancer, antinociceptive, cytotoxic, antihyperlipidemic, antispasmodic, and immunomodulatory effects [6], and is effective in treating headaches, congestion, and liver disorders [7]. It is known for its aromatic characteristics, which are attributed to its extract composition. The plant extract contains a significant amount of limonene, geranial, and other monoterpenes, which contribute to its flavor profile and potential medicinal properties [4].
Studies have shown that certain types of Melissa officinalis exhibit antibacterial, antifungal, antioxidant, antidiarrheal, and antidiuretic properties, and contain compounds such as rosmarinic acid, luteolin, and apigenin [8]. Dracocephalum kotschyi Boiss and Melissa officinalis belong to the mint family (Lamiaceae) and are cultivated in the Mediterranean region in a wide variety of different forms. The plant is native to Iran and can be found in areas such as Yasuj, Isfahan, Mazandaran, and Tabriz [5].
Heydari et al. assessed the effect of Melissa officinalis on depression in patients after coronary artery bypass surgery and concluded that the consumption of Melissa officinalis capsules for seven days significantly reduces depression in patients who had undergone coronary artery bypass surgery [6]. Kermanian et al., in their study comparing the effects of chamomile tea and black tea on depression and oxidative stress in type 2 diabetic patients, concluded that chamomile tea consumption leads to a relative reduction in depression but did not affect oxidative stress [9]. Hajrasouliha and Khakpour concluded that Melissa officinalis extract has an antidepressant effect similar to that of fluoxetine [10]. Najafi Doulatabad et al. reported that mint can be used as an adjunct in the treatment of depression [1]. Haybar et al. stated that lemon balm herb reduces depression, anxiety, and stress [11]. Ghazizadeh et al. demonstrated that the hydroalcoholic extract of Melissa officinalis has anti-anxiety and antidepressant effects in mice [12].
Although studies have been conducted on the effect of Melissa officinalis on depression in patients with various diseases, demonstrating its ability to reduce depression, no study has been found in which escitalopram was prescribed to both groups simultaneously, with the intervention group receiving Melissa officinalis in addition to escitalopram for comparison. In most studies, the control group was given a placebo alone. Furthermore, the Melissa officinalis from Dena may have different effects compared to Melissa officinalis from other locations. Therefore, considering the public’s inclination to use herbal medicines and the availability of the lemon balm plant [13], this study aimed to determine the effectiveness of consuming the hydroalcoholic extract from the lemon balm plant in patients with depression who are receiving escitalopram.
Materials and Methods
Study design, participants, and sampling
This two-group non-randomized controlled clinical trial was conducted on patients referred to Shahid Mofatteh Psychiatric Clinic in Yasuj during a one-year period from April to March 2023. The sample size was estimated using G*Power software based on the statistical requirements of the ANCOVA. The sample size calculation formula was based on an expected effect size of 0.67, a correlation coefficient between repeated measurements of 0.7, a type I error rate of 0.05, and a power of the test of 0.8. Considering the possibility of dropouts and lack of patient cooperation during the study, the sample size was determined to be 78 patients, divided into two groups (an intervention group and a control group) of 39 patients each. One group received escitalopram alone as the control group, while the other group received escitalopram along with a capsule of lemon balm extract as the intervention group.
The sample was collected using a non-random convenience sampling method (in an alternating manner, meaning that the first patient received escitalopram, the second patient received escitalopram along with lemon balm, the third patient received escitalopram again, and so on) from patients visiting the psychiatric clinic in Yasuj in the year 2023.
The inclusion criteria included a diagnosis of mild to moderate depression based on a clinical interview and the Beck Depression Inventory (with scores ranging from 10 to 29) and the Hamilton Depression Rating Scale (with scores ranging from 8 to 23), willingness to participate in the research, age between 15 and 65, absence of underlying diseases affecting depression, such as cancer or diabetes, and not having schizophrenia, schizoaffective disorder, psychosis, or anxiety disorders. Outcomes from the study included worsening of the disease, serious suicidal thoughts, non-cooperation during the study, failure to complete the questionnaire, and any general medical illness that occurred during the study and led to discontinuation (e.g., myocardial infarction).
Data collection tools
Data were collected using the Hamilton Depression Rating Scale and the Beck Depression Inventory.
The Hamilton Depression Rating Scale, designed to assess the severity of depressive symptoms through a structured clinical interview, was developed by Hamilton [14]. This scale consists of 24 items and is scored by Ballinger using the Likert scale. Its concurrent and construct validity have been independently confirmed through agreement between raters in HDRS scores and structured clinical interview scores. The sensitivity of the scale in detecting positive cases of depression is 87%. The highest sensitivity and specificity of HDRS in multiple studies have been observed at the clinical cutoff point of 17, where the sensitivity was 62.4% and the specificity was 92% [15]. In Iran, the validity of this instrument has been assessed through its correlation with the Beck Depression Inventory and the Dysfunctional Attitudes Scale, yielding coefficients of 55% and 39%, respectively. Additionally, the inter-rater reliability has been reported at 95%, indicating a high level of consistency in assessments [16]. Based on previous studies, scores ranging from 0 to 7 indicate the absence of depression, scores between 8 and 16 suggest mild depression, scores from 17 to 23 reflect moderate depression, and scores of 24 or higher are classified as severe depression [17].
The Beck Depression Inventory consists of 21 items, with each item scored between 0 and 3. The total score ranges from 0 to 63, indicating varying levels of depressive symptoms [15]. The concurrent validity of the questionnaire, determined through its correlation with a psychiatric interview, was found to be 77%. Additionally, its reliability was assessed using the split-half method, yielding a reliability coefficient of 90%, while the test-retest method produced a reliability coefficient of 75% [16]. Hojat et al. conducted a study to evaluate the validity and reliability of the Beck Depression Inventory among Iranian students residing in Iran and the United States. The findings revealed that the Cronbach’s alpha coefficient for both groups was 0.85 and 0.83, respectively, indicating a high level of reliability for the questionnaire. Furthermore, using the test-retest method after three weeks, a reliability coefficient of 0.61 was obtained, demonstrating acceptable reliability for the instrument [17]. According to the authors’ recommendations (Beck et al.), a score below 10 indicates minimal or no depression, a score between 10 and 18 suggests mild to moderate depression, a score between 19 and 29 reflects moderate to severe depression, and a score between 30 and 63 signifies severe depression [18].
Preparation of Melissa officinalis extract
The Melissa officinalis was collected during the summer from the Dena Mountain range surrounding Yasuj and transported to the Medicinal Plants Research Center at Yasuj University of Medical Sciences, where it was identified by a botany specialist. Various parts of the plant sample, including leaves, stems, and flowers, were then separated, dried in the shade at room temperature away from direct sunlight, and extracted using a hydroalcoholic method.
To prepare the hydroalcoholic extract, 100g of dried powder was mixed with 500mL of 70% ethanol and stored for 72 hours at room temperature (22°C) in a dark environment. The resulting extract was filtered using Whatman No. 1 filter paper and subjected to ethanol removal through a rotary evaporator (EV311 lab tech) operating at 100rpm and 40°C. The alcohol extract was then dried in an incubator at 40°C and stored in a refrigerator until the experiment was conducted. Finally, the powdered extract was encapsulated using a laboratory-grade filling machine to determine appropriate dosage levels [19].
Intervention method and data collection
After obtaining approval for the ethical code and necessary permits, this study was conducted on patients referred to the Shahid Mofatteh Psychiatric Clinic in Yasuj. Patients aged 15 to 65 years were interviewed by a psychiatrist, underwent psychiatric evaluations and clinical tests, and were included in the study if they were willing to participate. They entered the process with informed consent and were informed about the extract. First, the patients were interviewed by a psychiatrist; second, they underwent a psychiatric evaluation and clinical examination; and third, the Beck and Hamilton Depression Scales were completed on the first day of the study. In the control group, patients received a dose of 10mg of escitalopram tablets once a day for one month, while in the experimental group, patients received a daily dose of 500mg of lemon balm extract once a day for thirty days, in addition to taking 10mg of escitalopram tablets once a day. One month after the intervention, a re-evaluation was conducted by the interviewing psychiatrist, including a psychiatric examination, clinical examination, and completion of the Beck and Hamilton Depression Scales. Finally, the collected data were analyzed statistically.
Blinding
Although patients could not be blinded, the data gatherer and the statistician were blinded to the patients in the groups.
Statistical analysis
Collected data were analyzed using SPSS 19 software, with p<0.05 considered a statistically significant difference. Chi-square tests and independent sample t-tests were employed for demographic parameters, as the statistical distribution of the outcome parameters was normal. The independent samples t-test, dependent samples t-test, and paired samples t-test were used for between-group and within-group comparisons.
Findings
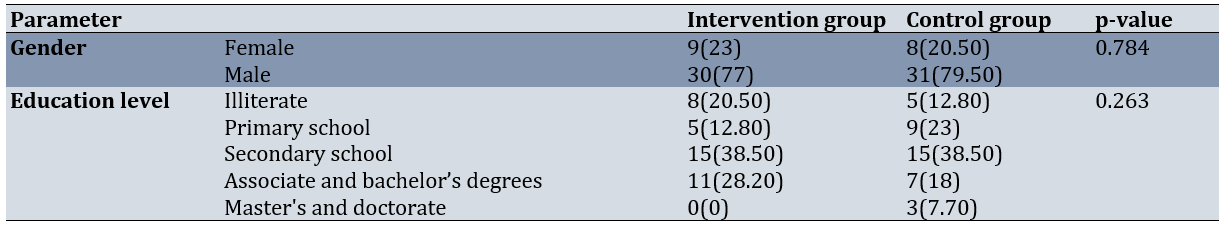
The Chi-square test indicated no significant difference between the two groups in terms of gender (p=0.784) or educational attainment (p=0.263). An independent t-test showed no significant difference in age between the intervention (35.38±12.90) and control (34.74±10.61) groups (p=0.811; Table 1).
Table 1. Frequency of demographic characteristics in the two studied groups
The independent t-test results demonstrated that the two groups did not differ significantly in pre- and post-test depression severity on the Beck Depression Inventory (p=0.533), but post-test depression severity was significantly reduced in both groups (p<0.001). The severity of depression on the Beck Depression Inventory was significantly lower in the intervention group (p<0.001; Table 2).
Table 2. Mean depression severity in the intervention and control groups

The independent t-test results indicated no significant difference between the two groups in terms of severity of depression on the Hamilton Depression Rating Scale prior to the intervention (p=0.570). Additionally, the paired t-test results demonstrated that the severity of depression on the Hamilton scale decreased significantly after the intervention (p<0.001). However, the severity of depression on the Hamilton scale in the intervention group was significantly lower (p<0.001; Table 3).
Table 3. Assessment and comparison of mean Hamilton depression severity in intervention and control groups

Discussion
This study aimed to determine the effectiveness of the hydroalcoholic extract from Melissa officinalis in depressed patients taking escitalopram. The results of this study showed that both the intervention group (using Melissa officinalis hydroalcoholic extract with escitalopram) and the control group (taking escitalopram alone) experienced an improvement in depressive symptoms after the intervention compared to before the intervention.
One of the characteristics of escitalopram that distinguishes it from other SSRIs is its allosteric property. This unique property allows it to modulate serotonin reuptake through mechanisms that extend beyond traditional serotonin reuptake inhibitors [20]. Moreover, the combination of escitalopram with other antidepressants, such as mirtazapine, has been shown to be beneficial by engaging different mechanisms of action and may improve treatment outcomes [21]. Here, a similar effect was observed when escitalopram was used alone; however, the combination of Melissa officinalis extract and escitalopram resulted in a more pronounced improvement in the depression of the patients.
Heidari et al. found that consuming Melissa officinalis capsules for seven days results in a significant reduction in depression levels among patients undergoing coronary artery bypass surgery. Fear of returning to work and normal life is a major factor in the development of depression in these patients. Therefore, this herbal medicine could be incorporated into the treatment regimen of these patients to prevent adverse postoperative and discharge outcomes [13]. This study is consistent with the findings of the aforementioned article, and based on the results obtained, Dena Melissa officinalis may be effective in reducing depression. The difference from the present study is that, in the above study, a placebo was used for the control group, whereas in the present study, escitalopram was administered to both groups, with Dena Melissa officinalis added to the intervention group.
In line with the present study, Hajrasouliha and Khakpour reported that the extract of Melissa officinalis has an antidepressant effect similar to that of fluoxetine [10]. This study aligns with the findings of the aforementioned article, and based on the results obtained, Melissa officinalis Denayi may have an effect on reducing depression. The difference between the two studies is that fluoxetine was compared with Melissa officinalis, and the study was conducted on animals. Furthermore, to evaluate the antidepressant effect of Melissa officinalis, the forced swimming test and the suspension test were employed in male Syrian mice.
Haybar et al. showed that the intervention group receiving Melissa officinalis capsules has a significant reduction in depression, anxiety, stress, and overall sleep disorder scores compared to the placebo group [11]. Similarly, as observed in the present study, the results indicated that Melissa officinalis plays a role in reducing the level of depression in patients. Moreover, it leads to an improvement in patients’ quality of life. The difference between this study and ours is that a placebo was administered to the control group, whereas in the present study, escitalopram was prescribed to both groups.
Additionally, as observed in the present study, Talebi et al. demonstrated that Melissa officinalis extract has an antidepressant effect [14]. Melissa officinalis is an important medicinal plant with a large number of active constituents and significant potential to safeguard human and animal health in Iran. The plant has also been reported to remedy inflammatory pain, headaches, congestion, fever, renal pain, dyspepsia, muscle spasms, and wound healing effects [6]. Dracocephalum kotschyi has anti-hyperlipidemic and anti-spasmodic activities and is effective in the treatment of headaches, congestion, and liver disorders [7].
Melissa officinalis was helpful as an auxiliary drug in the treatment of depression; our findings are somewhat consistent with these two studies. Melissa officinalis has anti-diabetic and anti-depressant activities. One study aimed to determine the efficacy of Melissa officinalis extract on depression, anxiety, and sleep quality in patients with type 2 diabetes who have depressive symptoms [22]. Although the aforementioned study has demonstrated the effect of Melissa officinalis on depression in patients with diabetes, and the present study showed the effect of this plant on depression in patients with depressive disorders, both studies observed an improvement in depression. Melissa officinalis (lemon balm) significantly improved mean anxiety and depression scores compared with the placebo, without serious side effects. Melissa officinalis may be effective in improving anxiety and depressive symptoms, particularly in acute settings [23].
While improvement in depression was noted in both studies, the difference between this study and ours is that the control group received a placebo and did not use any antidepressants, such as escitalopram or others. The intervention group received only Melissa officinalis.
The use of Melissa officinalis could reduce the incidence rate of postpartum blues, which is one of the most common postpartum psychiatric disorders, without the development of potential side effects [24]. Melissa officinalis has been shown to possess serotonergic antidepressant activity, which may help prevent depression in rats [25]. Additionally, Melissa officinalis can significantly reduce anxiety and depression, improve sleep quality, and may be beneficial for burn patients as a daily drink [26].
The studies mentioned have shown the effect of Melissa officinalis on the improvement of psychological problems after childbirth, depression, and anxiety in burn patients, and depression in rats. Although the nature of the patients’ depression differed and the sample sizes in these two studies were not the same, the results are somewhat consistent with those of the present study.
Although the strength of the study was the observance of research ethics and the administration of antidepressants to both groups, in addition to the intervention group, it also presented a limitation. This was due to the possibility of a synergistic effect, and the outcome may have been different if escitalopram had been prescribed to only one group. Another limitation of the study was the use of a mental health questionnaire, which may not accurately reflect the level of depression.
People have long recognized that plants can be used as remedies for certain diseases, and some plants are effective in treating neurological and psychiatric conditions. Melissa officinalis was found to be effective as an adjunctive treatment for mild to moderate depressive disorders. However, to confirm these results and to introduce Melissa officinalis as a new treatment option, more extensive and long-term clinical studies are needed to examine the appropriate dosage, precise mechanisms of action, and possible side effects of this plant. Additionally, if feasible, a study that includes an intervention group without escitalopram should be designed and implemented.
Conclusion
The extract of Melissa officinalis is effective in reducing symptoms of depression when used in combination with the medication escitalopram.
Acknowledgments: The authors would like to thank the participants of the study and Yasuj University of Medical Sciences for their collaboration in financing this research.
Ethical Permissions: This study was registered in the Iranian Registry of Clinical Trials with registration reference IRCT20240527061917N1. The study protocol was approved by the Ethics Committee of Yasuj University of Medical Sciences (Ethical code IR.YUMS.REC.1402.154). Furthermore, voluntary participation, the confidentiality of participants’ data, and the absence of financial burdens on participants were ensured.
Conflicts of Interests: The authors have no conflicts of interests to declare.
Authors' Contribution: Hashemimohammadabad Z (First Author), Introduction Writer/Main Researcher/Discussion Writer (20%); Hashemimohammadabad M (Second Author), Introduction Writer/Methodologist/Assistant Researcher/Discussion Writer (15%); Yazdanpanah I (Third Author), Introduction Writer/Assistant Researcher/Discussion Writer (10%); Razmjoue D (Fourth Author), Methodologist/Assistant Researcher/Statistical Analyst (15%); Karimi F (Fifth Author), Introduction Writer/Methodologist/Assistant Researcher/Discussion Writer (15%); Hashemimohammadabad N (Sixth Author), Introduction Writer/Methodologist/Main Researcher/Discussion Writer (25%)
Funding/Support: This article was extracted from Fatemeh Karimi's thesis in medicine and was supported by Yasuj University of Medical Sciences.
Depression is a common psychiatric disorder and one of the most disabling mental health issues in various societies [1]. It is characterized by a mental state of low mood and reduced activity, affecting over 280 million people (about 3.5% of the world’s population). Classified as a mental and behavioral disorder, depression impacts the way people think, act, and feel. The main symptoms of depression may include a lack of interest and enjoyment in life (anhedonia), a mood disorder, or both [2]. Depression is a serious condition in today’s society, with one in 10 patients suffering from major depression [3].
The most commonly prescribed antidepressants are selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and tricyclic antidepressants (TCAs). SSRIs are among the newer treatments for major depression and are typically used as a first-line treatment. Escitalopram is one of these medications, prescribed in daily doses ranging from 10 to 60mg [4].
Antidepressants can take several weeks to show effects and may be accompanied by side effects. Some patients may struggle to adhere to medication due to these side effects or fear of using chemicals, which can lead to ineffective treatment [3].
Plants have been an important source of medicine for thousands of years. The World Health Organization estimates that nearly 80% of people still primarily rely on traditional remedies, such as herbs, for their medicines. Plants are also the source of many modern medicines. It is estimated that up to one quarter of prescribed drugs contain plant extracts or active ingredients obtained from plant substances [5].
Dracocephalum kotschyi Boiss possesses a number of medicinal properties, including antioxidant, antibacterial, anticancer, antinociceptive, cytotoxic, antihyperlipidemic, antispasmodic, and immunomodulatory effects [6], and is effective in treating headaches, congestion, and liver disorders [7]. It is known for its aromatic characteristics, which are attributed to its extract composition. The plant extract contains a significant amount of limonene, geranial, and other monoterpenes, which contribute to its flavor profile and potential medicinal properties [4].
Studies have shown that certain types of Melissa officinalis exhibit antibacterial, antifungal, antioxidant, antidiarrheal, and antidiuretic properties, and contain compounds such as rosmarinic acid, luteolin, and apigenin [8]. Dracocephalum kotschyi Boiss and Melissa officinalis belong to the mint family (Lamiaceae) and are cultivated in the Mediterranean region in a wide variety of different forms. The plant is native to Iran and can be found in areas such as Yasuj, Isfahan, Mazandaran, and Tabriz [5].
Heydari et al. assessed the effect of Melissa officinalis on depression in patients after coronary artery bypass surgery and concluded that the consumption of Melissa officinalis capsules for seven days significantly reduces depression in patients who had undergone coronary artery bypass surgery [6]. Kermanian et al., in their study comparing the effects of chamomile tea and black tea on depression and oxidative stress in type 2 diabetic patients, concluded that chamomile tea consumption leads to a relative reduction in depression but did not affect oxidative stress [9]. Hajrasouliha and Khakpour concluded that Melissa officinalis extract has an antidepressant effect similar to that of fluoxetine [10]. Najafi Doulatabad et al. reported that mint can be used as an adjunct in the treatment of depression [1]. Haybar et al. stated that lemon balm herb reduces depression, anxiety, and stress [11]. Ghazizadeh et al. demonstrated that the hydroalcoholic extract of Melissa officinalis has anti-anxiety and antidepressant effects in mice [12].
Although studies have been conducted on the effect of Melissa officinalis on depression in patients with various diseases, demonstrating its ability to reduce depression, no study has been found in which escitalopram was prescribed to both groups simultaneously, with the intervention group receiving Melissa officinalis in addition to escitalopram for comparison. In most studies, the control group was given a placebo alone. Furthermore, the Melissa officinalis from Dena may have different effects compared to Melissa officinalis from other locations. Therefore, considering the public’s inclination to use herbal medicines and the availability of the lemon balm plant [13], this study aimed to determine the effectiveness of consuming the hydroalcoholic extract from the lemon balm plant in patients with depression who are receiving escitalopram.
Materials and Methods
Study design, participants, and sampling
This two-group non-randomized controlled clinical trial was conducted on patients referred to Shahid Mofatteh Psychiatric Clinic in Yasuj during a one-year period from April to March 2023. The sample size was estimated using G*Power software based on the statistical requirements of the ANCOVA. The sample size calculation formula was based on an expected effect size of 0.67, a correlation coefficient between repeated measurements of 0.7, a type I error rate of 0.05, and a power of the test of 0.8. Considering the possibility of dropouts and lack of patient cooperation during the study, the sample size was determined to be 78 patients, divided into two groups (an intervention group and a control group) of 39 patients each. One group received escitalopram alone as the control group, while the other group received escitalopram along with a capsule of lemon balm extract as the intervention group.
The sample was collected using a non-random convenience sampling method (in an alternating manner, meaning that the first patient received escitalopram, the second patient received escitalopram along with lemon balm, the third patient received escitalopram again, and so on) from patients visiting the psychiatric clinic in Yasuj in the year 2023.
The inclusion criteria included a diagnosis of mild to moderate depression based on a clinical interview and the Beck Depression Inventory (with scores ranging from 10 to 29) and the Hamilton Depression Rating Scale (with scores ranging from 8 to 23), willingness to participate in the research, age between 15 and 65, absence of underlying diseases affecting depression, such as cancer or diabetes, and not having schizophrenia, schizoaffective disorder, psychosis, or anxiety disorders. Outcomes from the study included worsening of the disease, serious suicidal thoughts, non-cooperation during the study, failure to complete the questionnaire, and any general medical illness that occurred during the study and led to discontinuation (e.g., myocardial infarction).
Data collection tools
Data were collected using the Hamilton Depression Rating Scale and the Beck Depression Inventory.
The Hamilton Depression Rating Scale, designed to assess the severity of depressive symptoms through a structured clinical interview, was developed by Hamilton [14]. This scale consists of 24 items and is scored by Ballinger using the Likert scale. Its concurrent and construct validity have been independently confirmed through agreement between raters in HDRS scores and structured clinical interview scores. The sensitivity of the scale in detecting positive cases of depression is 87%. The highest sensitivity and specificity of HDRS in multiple studies have been observed at the clinical cutoff point of 17, where the sensitivity was 62.4% and the specificity was 92% [15]. In Iran, the validity of this instrument has been assessed through its correlation with the Beck Depression Inventory and the Dysfunctional Attitudes Scale, yielding coefficients of 55% and 39%, respectively. Additionally, the inter-rater reliability has been reported at 95%, indicating a high level of consistency in assessments [16]. Based on previous studies, scores ranging from 0 to 7 indicate the absence of depression, scores between 8 and 16 suggest mild depression, scores from 17 to 23 reflect moderate depression, and scores of 24 or higher are classified as severe depression [17].
The Beck Depression Inventory consists of 21 items, with each item scored between 0 and 3. The total score ranges from 0 to 63, indicating varying levels of depressive symptoms [15]. The concurrent validity of the questionnaire, determined through its correlation with a psychiatric interview, was found to be 77%. Additionally, its reliability was assessed using the split-half method, yielding a reliability coefficient of 90%, while the test-retest method produced a reliability coefficient of 75% [16]. Hojat et al. conducted a study to evaluate the validity and reliability of the Beck Depression Inventory among Iranian students residing in Iran and the United States. The findings revealed that the Cronbach’s alpha coefficient for both groups was 0.85 and 0.83, respectively, indicating a high level of reliability for the questionnaire. Furthermore, using the test-retest method after three weeks, a reliability coefficient of 0.61 was obtained, demonstrating acceptable reliability for the instrument [17]. According to the authors’ recommendations (Beck et al.), a score below 10 indicates minimal or no depression, a score between 10 and 18 suggests mild to moderate depression, a score between 19 and 29 reflects moderate to severe depression, and a score between 30 and 63 signifies severe depression [18].
Preparation of Melissa officinalis extract
The Melissa officinalis was collected during the summer from the Dena Mountain range surrounding Yasuj and transported to the Medicinal Plants Research Center at Yasuj University of Medical Sciences, where it was identified by a botany specialist. Various parts of the plant sample, including leaves, stems, and flowers, were then separated, dried in the shade at room temperature away from direct sunlight, and extracted using a hydroalcoholic method.
To prepare the hydroalcoholic extract, 100g of dried powder was mixed with 500mL of 70% ethanol and stored for 72 hours at room temperature (22°C) in a dark environment. The resulting extract was filtered using Whatman No. 1 filter paper and subjected to ethanol removal through a rotary evaporator (EV311 lab tech) operating at 100rpm and 40°C. The alcohol extract was then dried in an incubator at 40°C and stored in a refrigerator until the experiment was conducted. Finally, the powdered extract was encapsulated using a laboratory-grade filling machine to determine appropriate dosage levels [19].
Intervention method and data collection
After obtaining approval for the ethical code and necessary permits, this study was conducted on patients referred to the Shahid Mofatteh Psychiatric Clinic in Yasuj. Patients aged 15 to 65 years were interviewed by a psychiatrist, underwent psychiatric evaluations and clinical tests, and were included in the study if they were willing to participate. They entered the process with informed consent and were informed about the extract. First, the patients were interviewed by a psychiatrist; second, they underwent a psychiatric evaluation and clinical examination; and third, the Beck and Hamilton Depression Scales were completed on the first day of the study. In the control group, patients received a dose of 10mg of escitalopram tablets once a day for one month, while in the experimental group, patients received a daily dose of 500mg of lemon balm extract once a day for thirty days, in addition to taking 10mg of escitalopram tablets once a day. One month after the intervention, a re-evaluation was conducted by the interviewing psychiatrist, including a psychiatric examination, clinical examination, and completion of the Beck and Hamilton Depression Scales. Finally, the collected data were analyzed statistically.
Blinding
Although patients could not be blinded, the data gatherer and the statistician were blinded to the patients in the groups.
Statistical analysis
Collected data were analyzed using SPSS 19 software, with p<0.05 considered a statistically significant difference. Chi-square tests and independent sample t-tests were employed for demographic parameters, as the statistical distribution of the outcome parameters was normal. The independent samples t-test, dependent samples t-test, and paired samples t-test were used for between-group and within-group comparisons.
Findings
The Chi-square test indicated no significant difference between the two groups in terms of gender (p=0.784) or educational attainment (p=0.263). An independent t-test showed no significant difference in age between the intervention (35.38±12.90) and control (34.74±10.61) groups (p=0.811; Table 1).
Table 1. Frequency of demographic characteristics in the two studied groups

The independent t-test results demonstrated that the two groups did not differ significantly in pre- and post-test depression severity on the Beck Depression Inventory (p=0.533), but post-test depression severity was significantly reduced in both groups (p<0.001). The severity of depression on the Beck Depression Inventory was significantly lower in the intervention group (p<0.001; Table 2).
Table 2. Mean depression severity in the intervention and control groups

The independent t-test results indicated no significant difference between the two groups in terms of severity of depression on the Hamilton Depression Rating Scale prior to the intervention (p=0.570). Additionally, the paired t-test results demonstrated that the severity of depression on the Hamilton scale decreased significantly after the intervention (p<0.001). However, the severity of depression on the Hamilton scale in the intervention group was significantly lower (p<0.001; Table 3).
Table 3. Assessment and comparison of mean Hamilton depression severity in intervention and control groups

Discussion
This study aimed to determine the effectiveness of the hydroalcoholic extract from Melissa officinalis in depressed patients taking escitalopram. The results of this study showed that both the intervention group (using Melissa officinalis hydroalcoholic extract with escitalopram) and the control group (taking escitalopram alone) experienced an improvement in depressive symptoms after the intervention compared to before the intervention.
One of the characteristics of escitalopram that distinguishes it from other SSRIs is its allosteric property. This unique property allows it to modulate serotonin reuptake through mechanisms that extend beyond traditional serotonin reuptake inhibitors [20]. Moreover, the combination of escitalopram with other antidepressants, such as mirtazapine, has been shown to be beneficial by engaging different mechanisms of action and may improve treatment outcomes [21]. Here, a similar effect was observed when escitalopram was used alone; however, the combination of Melissa officinalis extract and escitalopram resulted in a more pronounced improvement in the depression of the patients.
Heidari et al. found that consuming Melissa officinalis capsules for seven days results in a significant reduction in depression levels among patients undergoing coronary artery bypass surgery. Fear of returning to work and normal life is a major factor in the development of depression in these patients. Therefore, this herbal medicine could be incorporated into the treatment regimen of these patients to prevent adverse postoperative and discharge outcomes [13]. This study is consistent with the findings of the aforementioned article, and based on the results obtained, Dena Melissa officinalis may be effective in reducing depression. The difference from the present study is that, in the above study, a placebo was used for the control group, whereas in the present study, escitalopram was administered to both groups, with Dena Melissa officinalis added to the intervention group.
In line with the present study, Hajrasouliha and Khakpour reported that the extract of Melissa officinalis has an antidepressant effect similar to that of fluoxetine [10]. This study aligns with the findings of the aforementioned article, and based on the results obtained, Melissa officinalis Denayi may have an effect on reducing depression. The difference between the two studies is that fluoxetine was compared with Melissa officinalis, and the study was conducted on animals. Furthermore, to evaluate the antidepressant effect of Melissa officinalis, the forced swimming test and the suspension test were employed in male Syrian mice.
Haybar et al. showed that the intervention group receiving Melissa officinalis capsules has a significant reduction in depression, anxiety, stress, and overall sleep disorder scores compared to the placebo group [11]. Similarly, as observed in the present study, the results indicated that Melissa officinalis plays a role in reducing the level of depression in patients. Moreover, it leads to an improvement in patients’ quality of life. The difference between this study and ours is that a placebo was administered to the control group, whereas in the present study, escitalopram was prescribed to both groups.
Additionally, as observed in the present study, Talebi et al. demonstrated that Melissa officinalis extract has an antidepressant effect [14]. Melissa officinalis is an important medicinal plant with a large number of active constituents and significant potential to safeguard human and animal health in Iran. The plant has also been reported to remedy inflammatory pain, headaches, congestion, fever, renal pain, dyspepsia, muscle spasms, and wound healing effects [6]. Dracocephalum kotschyi has anti-hyperlipidemic and anti-spasmodic activities and is effective in the treatment of headaches, congestion, and liver disorders [7].
Melissa officinalis was helpful as an auxiliary drug in the treatment of depression; our findings are somewhat consistent with these two studies. Melissa officinalis has anti-diabetic and anti-depressant activities. One study aimed to determine the efficacy of Melissa officinalis extract on depression, anxiety, and sleep quality in patients with type 2 diabetes who have depressive symptoms [22]. Although the aforementioned study has demonstrated the effect of Melissa officinalis on depression in patients with diabetes, and the present study showed the effect of this plant on depression in patients with depressive disorders, both studies observed an improvement in depression. Melissa officinalis (lemon balm) significantly improved mean anxiety and depression scores compared with the placebo, without serious side effects. Melissa officinalis may be effective in improving anxiety and depressive symptoms, particularly in acute settings [23].
While improvement in depression was noted in both studies, the difference between this study and ours is that the control group received a placebo and did not use any antidepressants, such as escitalopram or others. The intervention group received only Melissa officinalis.
The use of Melissa officinalis could reduce the incidence rate of postpartum blues, which is one of the most common postpartum psychiatric disorders, without the development of potential side effects [24]. Melissa officinalis has been shown to possess serotonergic antidepressant activity, which may help prevent depression in rats [25]. Additionally, Melissa officinalis can significantly reduce anxiety and depression, improve sleep quality, and may be beneficial for burn patients as a daily drink [26].
The studies mentioned have shown the effect of Melissa officinalis on the improvement of psychological problems after childbirth, depression, and anxiety in burn patients, and depression in rats. Although the nature of the patients’ depression differed and the sample sizes in these two studies were not the same, the results are somewhat consistent with those of the present study.
Although the strength of the study was the observance of research ethics and the administration of antidepressants to both groups, in addition to the intervention group, it also presented a limitation. This was due to the possibility of a synergistic effect, and the outcome may have been different if escitalopram had been prescribed to only one group. Another limitation of the study was the use of a mental health questionnaire, which may not accurately reflect the level of depression.
People have long recognized that plants can be used as remedies for certain diseases, and some plants are effective in treating neurological and psychiatric conditions. Melissa officinalis was found to be effective as an adjunctive treatment for mild to moderate depressive disorders. However, to confirm these results and to introduce Melissa officinalis as a new treatment option, more extensive and long-term clinical studies are needed to examine the appropriate dosage, precise mechanisms of action, and possible side effects of this plant. Additionally, if feasible, a study that includes an intervention group without escitalopram should be designed and implemented.
Conclusion
The extract of Melissa officinalis is effective in reducing symptoms of depression when used in combination with the medication escitalopram.
Acknowledgments: The authors would like to thank the participants of the study and Yasuj University of Medical Sciences for their collaboration in financing this research.
Ethical Permissions: This study was registered in the Iranian Registry of Clinical Trials with registration reference IRCT20240527061917N1. The study protocol was approved by the Ethics Committee of Yasuj University of Medical Sciences (Ethical code IR.YUMS.REC.1402.154). Furthermore, voluntary participation, the confidentiality of participants’ data, and the absence of financial burdens on participants were ensured.
Conflicts of Interests: The authors have no conflicts of interests to declare.
Authors' Contribution: Hashemimohammadabad Z (First Author), Introduction Writer/Main Researcher/Discussion Writer (20%); Hashemimohammadabad M (Second Author), Introduction Writer/Methodologist/Assistant Researcher/Discussion Writer (15%); Yazdanpanah I (Third Author), Introduction Writer/Assistant Researcher/Discussion Writer (10%); Razmjoue D (Fourth Author), Methodologist/Assistant Researcher/Statistical Analyst (15%); Karimi F (Fifth Author), Introduction Writer/Methodologist/Assistant Researcher/Discussion Writer (15%); Hashemimohammadabad N (Sixth Author), Introduction Writer/Methodologist/Main Researcher/Discussion Writer (25%)
Funding/Support: This article was extracted from Fatemeh Karimi's thesis in medicine and was supported by Yasuj University of Medical Sciences.
Keywords:
References
1. Najafi Doulatabad S, Hashemi N, Mohebi Nobandegani Z, Sadeghi H, Jafari A, Najafi Doulatabad A, et al. The effect of hydroalcoholic extract of mentha piperita (complement treatment) on severity of depressed patients. ARMAGHAN-E-DANESH. 2009;14(3):83-90. [Persian] [Link]
2. Fornells-Ambrojo M, Craig T, Garety P. Occupational functioning in early non-affective psychosis: The role of attributional biases, symptoms and executive functioning. Epidemiol Psychiatr Sci. 2013;23(1):71-84. [Link] [DOI:10.1017/S2045796013000103]
3. Whooley MA, Simon GE. Managing depression in medical outpatients. N Engl J Med. 2000;343(26):1942-50. [Link] [DOI:10.1056/NEJM200012283432607]
4. Raggi A, Serretti A, Ferri R. A comprehensive overview of post-stroke depression treatment options. Int Clin Psychopharmacol. 2024;39(3):127-38. [Link] [DOI:10.1097/YIC.0000000000000532]
5. Tripathi L, Tripathi JN. Role of biotechnology in medicinal plants. Trop J Pharm Res. 2003;2(2):243-53. [Link] [DOI:10.4314/tjpr.v2i2.14607]
6. Heydari P, Yavari M, Adibi P, Asghari G, Ghanadian SM, Dida GO, et al. Medicinal properties and active constituents of Dracocephalum kotschyi and its significance in Iran: A systematic review. Evid Based Complement Altern Med. 2019;2019(1):9465309. [Link] [DOI:10.1155/2019/9465309]
7. Foroozandeh E, Asadi Gharooneh HA. Dracocephalum kotschyi Boiss: An Iranian endemic medicinal plant; A review. Journal of Medicinal Herbs. 2021;12(1):9-17. [Link]
8. De Smet PA, Nolen WA. St John's wort as an antidepressant. BMJ. 1996;313(7052):241-2. [Link] [DOI:10.1136/bmj.313.7052.241]
9. Kermanian S, Mozaffari Khosravi H, Zavar Reza J, Dastgerdi G, Lotfi M. A Comparison of the effect of chamomile tea and black tea consumption on depression and oxidative stress markers in depressed patients with type II diabetes: A randomized clinical trial. J TOLOOEBEHDASHT. 2018;17(4):13-29. [Link] [DOI:10.18502/tbj.v17i4.183]
10. Hajrasouliha S, Khakpour S. Comparison of antidepressant effect of Melissa officinalis L. hydroalcoholic extract with fluoxetine in male mice. Med Sci. 2020;30(4):418-24. [Persian] [Link] [DOI:10.29252/iau.30.4.418]
11. Haybar H, Javid AZ, Haghighizadeh MH, Valizadeh E, Mohaghegh SM, Mohammadzadeh A. The effects of Melissa officinalis supplementation on depression, anxiety, stress, and sleep disorder in patients with chronic stable angina. Clin Nutr ESPEN. 2018;26:47-52. [Link] [DOI:10.1016/j.clnesp.2018.04.015]
12. Ghazizadeh J, Hamedeyazdan S, Torbati M, Farajdokht F, Fakhari A, Mahmoudi J, et al. Melissa officinalis L. hydro‐alcoholic extract inhibits anxiety and depression through prevention of central oxidative stress and apoptosis. Exp Physiol. 2020;105(4):707-20. [Link] [DOI:10.1113/EP088254]
13. Heidari M, Soltanpour A, Naseri M, Kazemnezhad A. The effect of Lemon Balm (Melissa Officinalis) on depression in patients after coronary artery bypass graft. Iran J Cardiovasc Nurs. 2015;4(2):36-43. [Persian] [Link]
14. Talebi S, Rahmati B, Jorjani M, Emadi F, Ghaffari F, Naseri M. Synergistic effects of Nepeta menthoides and Melissa officinalis aqueous extracts on reserpine‐induced depressive‐like behaviors in mice. Phytother Res. 2022;36(6):2481-94. [Link] [DOI:10.1002/ptr.7457]
15. Beck AT, Steer RA, Brown GK. Beck depression inventory-II. Washington DC: APA PsycTests; 1996. [Link] [DOI:10.1037/t00742-000]
16. Dozois DJ, Dobson KS, Ahnberg JL. A psychometric evaluation of the Beck Depression Inventory-II. Psychol Assess. 1998;10(2):83-9. [Link] [DOI:10.1037/1040-3590.10.2.83]
17. Hojat M, Shapurian R, Mehryar AH. Psychometric properties of a Persian version of the short form of the Beck Depression Inventory for Iranian college students. Psychol Rep. 1986;59(1):331-8. [Link] [DOI:10.2466/pr0.1986.59.1.331]
18. Beck AT, Steer RA, Carbin MG. Psychometric properties of the Beck Depression Inventory: Twenty-five years of evaluation. Clin Psychol Rev. 1988;8(1):77-100. [Link] [DOI:10.1016/0272-7358(88)90050-5]
19. Zargar A, Ardeshiri M, Khosravi A, Taheri Mirghaed A, Akbarein H, Ahmadpour M, et al. Study of in-vitro antimicrobial effects of Origanum vulgare and echinacea purpurea essential oils on Lactococcus garvieae. J Vet Res. 2023;77(4):213-20. [Persian] [Link]
20. Sanchez C, Reines EH, Montgomery SA. A comparative review of escitalopram, paroxetine, and sertraline: Are they all alike?. Int Clin Psychopharmacol. 2014;29(4):185-96. [Link] [DOI:10.1097/YIC.0000000000000023]
21. Himmerich H, Wranik DW. Choice of treatment with antidepressants: Influencing factors. Curr Pharm Des. 2012;18(36):5958-75. [Link] [DOI:10.2174/138161212803523653]
22. Safari M, Asadi A, Aryaeian N, Huseini HF, Shidfar F, Jazayeri S, et al. The effects of melissa officinalis on depression and anxiety in type 2 diabetes patients with depression: A randomized double-blinded placebo-controlled clinical trial. BMC Complement Med Ther. 2023;23(1):140. [Link] [DOI:10.1186/s12906-023-03978-x]
23. Ghazizadeh J, Sadigh‐Eteghad S, Marx W, Fakhari A, Hamedeyazdan S, Torbati M, et al. The effects of lemon balm (Melissa officinalis L.) on depression and anxiety in clinical trials: A systematic review and meta‐analysis. Phytother Res. 2021;35(12):6690-705. [Link] [DOI:10.1002/ptr.7252]
24. Beihaghi M, Yousefzade S, Mazloom SR, Gharavi MM, Hamedi SS. The effect of Melissa officinalis on postpartum blues in women undergoing cesarean section. J Midwifery Reprod Health. 2019;7(2):1656-63. [Link]
25. Lin SH, Chou ML, Chen WC, Lai YS, Lu KH, Hao CW, et al. A medicinal herb, Melissa officinalis L. ameliorates depressive-like behavior of rats in the forced swimming test via regulating the serotonergic neurotransmitter. J Ethnopharmacol. 2015;175:266-72. [Link] [DOI:10.1016/j.jep.2015.09.018]
26. Chehroudi S, Fatemi MJ, Isfeedvajani MS, Salehi SH, Akbari H, Samimi R. Effects of Melissa officinalis L. on reducing stress, alleviating anxiety disorders, depression, and insomnia, and increasing total antioxidants in burn patients. Trauma Mon. 2017;22(4):7. [Link] [DOI:10.5812/traumamon.33630]








